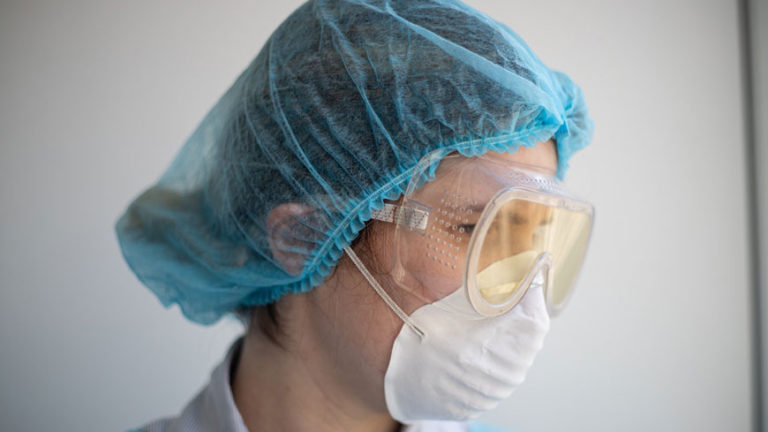
USP 797 – Garbing and Donning
View USP 797 gowning standards and core requirements. For sterile processing, additional precaution is needed to assure that no sterile surfaces contact non-sterile surfaces during gowning, processing, or cleaning. Sterile garments and gloves are recommended during aseptic workflows.

Aseptic Processing Guidelines – Most Common FDA Inspection Notes
The majority of contamination within aseptic processing cleanrooms involves personnel. Proper application of gowns, hygiene, and a work flow can often eliminate the majority of mix ups and contamination.

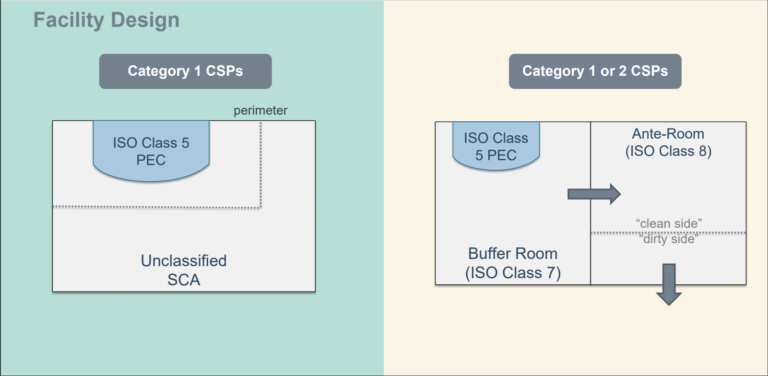
Gowning Room Design – Guidelines, Anterooms, and ISO Classification
A gowning room has many variables. There is no mathematical formula when determining the features, surfaces, and HVAC requirements best suited for your application. Space requirements, volume of operators, aseptic workflows, material choice, and even aesthetics all factor into design considerations. Most facilities benefit from contacting a controlled environment specialist who can identify critical product sensitivities, storage requirements, and operational standards.

Sterile Plant Tissue Culture Requirements: Environments, Chemicals, Garments
What do I need to tissue culture plants? What are the advantages of tissue culture? This article provides basic environment design, supplies, and protective requirements for non-hazardous sterile processes during the cell culturing and micro-propagation of clones. Learn about the tools and equipment required for aseptic workflow during tissue culture processes.

Kimberly-Clark Professional™ PPE, Garments, and Recycling Program (RightCycle)
Demand for eco-sensitive products and zero-waste initiatives has never been more pronounced. When you purchase Kimtech and KleenGuard products from Production Automation (PAC), you get the benefit of RightCycle by Kimberly-Clark Professional. This program delivers a sustainable approach for managing used nitrile gloves, single-use apparel items, and safety eyewear.

Particulate Matter – What is it and what are the health effects?
Historically, air quality has shown a complex relation to respiratory issues, birth defects, chronic illness, cancer, and even loss of cognitive function. Research suggests air pollution affects everything from neurodevelopment in-utero to cognitive decline in older people.

2019 Cleanroom Trends: Static Control, Process Monitoring, and Quality Control
Trends in the cleanroom market: reducing production costs, improving environmental conditions, and extending customer support with expertise.

New: Texwipe Cleanroom Products
Texwipe offers a variety of dry, pre-saturated, and sterile wipes to meet the strict demands of controlled environments. Texwipe swabs are manufactured to exacting and consistent tolerances with lot coded for traceability and quality control.

Proper Material for ESD Cleanroom Gloves
ESD cleanroom glove materials have reduced NVR (non-volatile residues) levels for better electrical conductivity. Meanwhile, dedicated ESD fabrics are often not viable for cleanroom use, as they tend to shed particulates at a much higher rate than latex, nitrile, or vinyl gloves.
Immunology and Microbiology Cleanrooms
Large clinical-scale cleanroom operations for T cell therapies often use a class ISO 7 (Class 10,000) cleanroom room environment with an ISO 5 (Class 100) biological safety hood.
Medical Device Cleanroom Classification
What Kind of Cleanroom Do I Need for a Class II Medical Device? What About Class III? This post classifies medical device cleanrooms for manufacturing and packaging.

New: Low Particulate Cleanroom Tape
Premium cleanroom tape and labels are feature non-particulating materials with plastic cores suitable for use in ISO 3 (Class 1) cleanrooms. Each roll and label is cleanroom bagged and packaged in a Class 100 (ISO Class 5) cleanroom to ensure cleanliness upon arrival.
Advantages of Peracetic Acid Sporicides
What are the advantages of peracetic acid sterilants for cleanroom disinfection? Where is it used? How does it compare to gas sterilization methods?

Cleanroom Fan Filter Replacement
When Should I Replace a Cleanroom Fan Filter? Pre-Filter Replacement Pre-filter media should be replaced every six months and inspected regularly. Pre-filters in gowning rooms

Guide to USP <1072> Disinfectants & Sporicides
USP <1072> outlines decontamination practices for critical environments.In this post, we’ll outline the types of chemical disinfectants and sterilants used in cleanrooms and laboratories. This includes isopropyl alcohol, bleach, formaldehyde, hydrogen peroxide, and peracetic acid.

ISO 7 Cleanroom Panel Cleaning & Maintenance
Warm water and a mild dish soap is the best softwall curtain cleaning solution. What about acrylic, melamine, and aluminum? Generally, any biodegradable solution is acceptable. A 50/50 solution of Isopropyl Alcohol & DI water is also effective. Wash curtains front side and back, rinse with clean water, then wipe dry with a clean lint-free soft cloth.

Cleanroom Fan Filter Units vs Air Handling Units
A cleanroom air handling unit (AHU) is a cornerstone of temperature, humidity, and pressurization within controlled environments. Today, modular fan filter units allow more adaptable configuration with simplified maintenance procedure. Both systems serve conditioned and filtered air throughout a cleanroom facility.

Air Ionization for Particulate & Contamination Control PDF from Simco-Ion
Our friends at Simco-Ion sent us this brilliant guide to ESD cleanrooms design, principles, and other considerations. Download the PDF you’ll find case studies and technical information about how static charges influence particle retention and impact manufacturing outcomes in controlled environments. Below we’ve highlighted important ideas in relation to medical device cleanrooms and provide additional resources.

Cleanrooms for Satellite & Spacecraft Construction
For communication satellites that remain within earth’s orbit, an ISO Class 6-8 cleanroom allows sufficient particle and contaminant control. Cleanroom engineers design each space so that particles from production surfaces are whisked away from critical components. A combination of softwall production areas and hardwall cleanroom facilities provide isolation of critical tasks.

Updated: USP Sterile Compounding 2019
First published in 2004, USP Chapter <797> has undergone proposed revisions as of July 2018. These revisions are now available for public comment.

