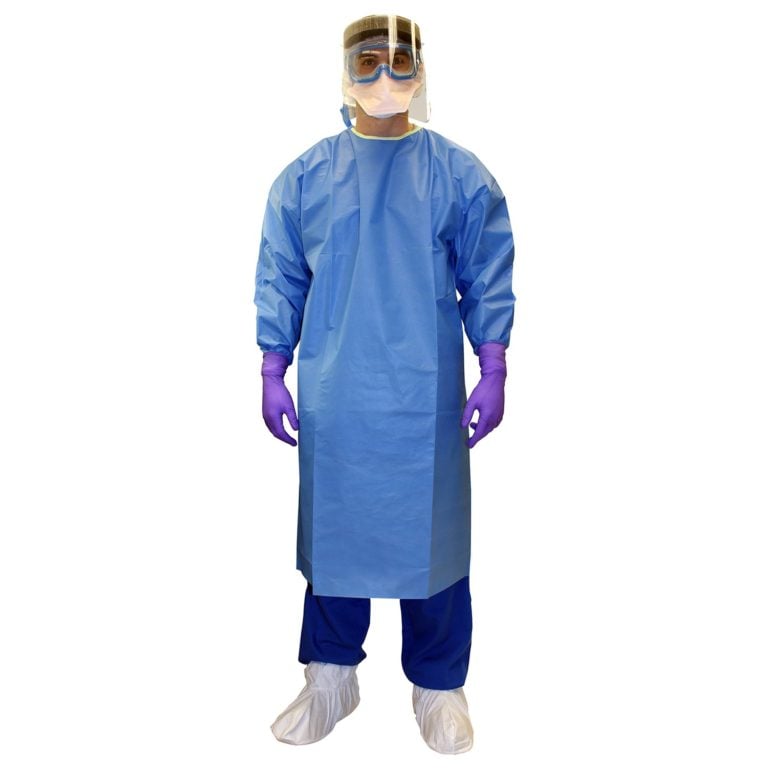
USP 800 Hazardous Garments
Appropriate PPE must be worn when handling HDs including during:
- Receipt
- Storage
- Transport
- Compounding (sterile and nonsterile)
- Administration
- Deactivation/decontamination, cleaning, and disinfecting
- Spill control
- Waste disposal
Summarized Requirements for USP 800 Gowning [source]
- Compounding of chemotherapy or hazardous drugs require chemotherapy gowns and chemotherapy rated gloves.
- Chemotherapy gloves are to be powder free, changed every 30 minutes, and thoroughly inspected before each use
- Gowns must be disposed after use, close in back, have no front opening, and have closed cuffs that are elastic or knit.
- Gowns should be replaced as required by governing body, after splashes and every 2 – 3 hours.
- An additional pair of shoe covers is required when entering secondary areas into HD handling areas
- Gloves must extend over the cuff of the gown and meet standards for the American Society for Testing and Materials (ASTM) Standard D6978 or later
When Are Chemotherapy Gloves Required?
Handling of all hazardous drugs (HDs), including non antineoplastics, and any HD with reproductive risks requires chemotherapy gloves. The standard for chemotherapy gloves is defined under the American Society for Testing and Materials (ASTM) standard D6978.
Chemotherapy Glove Requirements
- Always powder free.
- High integrity, no pin holes.
- Meets ASTM Standard D6978
- Double gloving determined by CSP risk factors
Chemotherapy Glove Standards
- Must be inspected for physical defects before use
- Sterile HD compounding requires sterile chemotherapy gloves
- Change gloves at least every 30 minutes or by manufacturers recommendation
- Changed after any tears, punctures, or contamination.
Gowns for Hazardous Drugs and Chemicals
Gowns for HD handling require chemical resistance and non-permeability after HD exposure. Permeability tests and standards are covered by ASTM F739-12. All gown manufacturers should provide results of ASTM F739-12 for certification and validation purposes.
Solution: One of our most commonly suggested hazardous compounding gowns is the Kimtech A7. Its liquid barrier is certified for compatibility with USP 800 hazardous compounding.
One of our most commonly suggested hazardous compounding gowns is the Kimtech A7. Its liquid barrier is certified for compatibility with USP 800 hazardous compounding.
Proper Procedure for Liquid Barrier Gowning and Removal
Not all cleanroom garments are made equal. One reason that many facilities favor Kimberly Clark garments is because they provide some unique features which make donning and gowning more simple. For example, color markings on garments help the operator remove the packaging from the garment with fewer variables, and reduce the risk of the operator transferring contamination.
“The material offers enhanced liquid barrier protection- fabric tested for blood and blood-borne pathogen penetration, chemotherapy drug and hormone permeation (passes ASTM F1670 for penetration of blood and body fluids and ASTM F1671 for blood borne pathogens) and the built-in thumb loops help maintain glove and garment interface. The gowns are low lint to minimize contamination.”
Disposable sleeve covers may be used to protect areas of the arm that may come in contact with HDs. Disposable sleeve covers made of polyethylene-coated polypropylene or other laminate materials offer better protection than those made of undercoated materials.
The proper cleanroom gowning room design, gowning supplies and equipment are an essential component of cleanroom contamination control. This informational guide should never take precedence over consulting with a cleanroom contamination specialist who is familiar with your specific equipment and classification needs. For help with cleanroom construction, gowning selection, or furniture purchases, our controlled environment specialists are available for a zero-cost consultation by phone or email.
USP <800> Certified Gowns
Overview of the Kimtech A7
- Thumb loops maintain glove and garment interface; preferred among compounding pharmacists.
- Seamless front and closed back; Easy to don and doff design
- Enhanced liquid barrier protection- fabric tested for blood and blood-borne pathogen penetration, chemotherapy drug and hormone permeation (passes ASTM F1670 for penetration of blood and body fluids) to help protect people and processes from liquid splashes
Find Expertise
Find a Specialist
Free ConsultationCompare Garments
Shop Garments
Shop USP PPEFind Solutions
USP Solutions
View OnlineRelated Posts
-
USP <800> Compounding Cleanrooms
Updated: USP 800 is a cleanroom standard issued in March of 2014 by the United States Pharmacopeial Convention (USP). The deadline December 2019 for compliance may change.
-
USP <800> Compounding Cleanrooms
Updated: USP 800 is a cleanroom standard issued in March of 2014 by the United States Pharmacopeial Convention (USP). The deadline December 2019 for compliance may change.
-
How Does USP 800 Change Storage and Unpacking of Hazardous Drugs?
Facilities unpacking hazardous drugs within a negative pressure room do not require procedural changes to meet USP unpacking area requirements. Facilities unpacking HDs within positive pressure ante rooms or facility spaces will require reconsideration.
-
Upgrading a USP 797 Cleanroom to USP 800?
New USP guidelines may present challenges for compounding facilities. Some facilities need infrastructural and mechanical modifications for compliance. System evaluation includes duct systems, HEPA fan filters, differential pressure standards, air monitoring, and external air exhaust…
-
CleanPro® Chemotherapy & CSP Cleanroom Installation
This modular, sterile compounding cleanroom is designed for USP 797 and USP 800 compliance, particularly for compounding chemotherapy drugs. Safe handling of sterile compounds requires special considerations: heat-welded floors, anterooms and buffer areas.
-
Class 10,000 Cleanroom & Gowning Area
This cleanroom and adjacent gowning area helped this customer prevent outer contamination from entering classified spaces.