
Cleanroom Fan Filter Replacement
When Should I Replace a Cleanroom Fan Filter? Pre-Filter Replacement Pre-filter media should be replaced every six months and inspected regularly. Pre-filters in gowning rooms

When Should I Replace a Cleanroom Fan Filter? Pre-Filter Replacement Pre-filter media should be replaced every six months and inspected regularly. Pre-filters in gowning rooms

USP <1072> outlines decontamination practices for critical environments.In this post, we’ll outline the types of chemical disinfectants and sterilants used in cleanrooms and laboratories. This includes isopropyl alcohol, bleach, formaldehyde, hydrogen peroxide, and peracetic acid.

Get a close up look at high-quality microscopes, LED magnifiers, and inspection devices from View Solutions.

Warm water and a mild dish soap is the best softwall curtain cleaning solution. What about acrylic, melamine, and aluminum? Generally, any biodegradable solution is acceptable. A 50/50 solution of Isopropyl Alcohol & DI water is also effective. Wash curtains front side and back, rinse with clean water, then wipe dry with a clean lint-free soft cloth.

A cleanroom air handling unit (AHU) is a cornerstone of temperature, humidity, and pressurization within controlled environments. Today, modular fan filter units allow more adaptable configuration with simplified maintenance procedure. Both systems serve conditioned and filtered air throughout a cleanroom facility.

Does a HEPA filter capture ultrafine particles (UFPs). What toxins does 3D printing produce? Why should every 3D printer have both a activated carbon and HEPA filter?

When should you choose stainless steel over chrome? Learn more about Metro wire shelving finishes including black, Metroseal®, MetroMax, and solid steel shelves.

M+A Matting produces an advanced version of the wiper mat called the Microluxx™. The surface excels at trapping and absorbing high levels of debris and moisture, retaining 2.5x more moisture than standard carpeted mats.

For communication satellites that remain within earth’s orbit, an ISO Class 6-8 cleanroom allows sufficient particle and contaminant control. Cleanroom engineers design each space so that particles from production surfaces are whisked away from critical components. A combination of softwall production areas and hardwall cleanroom facilities provide isolation of critical tasks.

View the Metro shelving accessories buyers guide. We’ve compiled the entire collection of Metro accessories onto one page for your convenience.
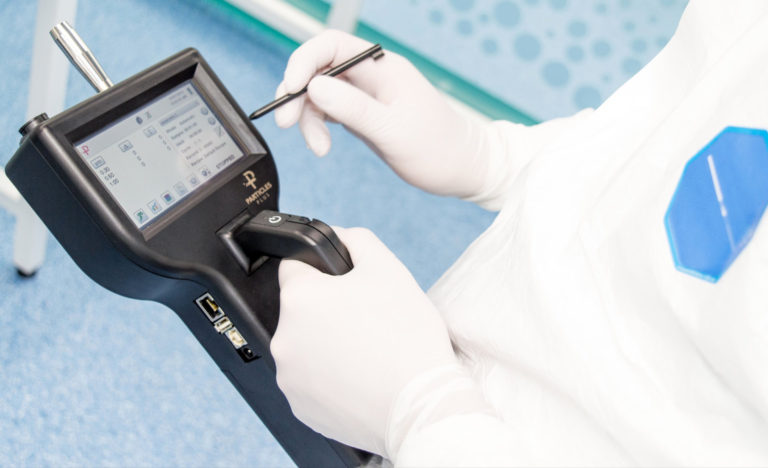
For ISO 14644 compliance, all particle counting equipment requires a calibration certificate.

Are you in compliance? Do you qualify for an exemption? Generic signage no longer adheres to recent Proposition 65 updates. Custom order Accuform signage is now available from PAC. Our specialists tailor hazard signage for your storage, entrance, and production areas.

In 1986, California voters addressed growing concerns over toxic chemical exposure. The resulting initiative was coined the Safe Drinking Water and Toxic enforcement as of 1986, commonly referred to as Proposition 65.

Aven’s newest magnifying lamp, In-X, stands for Interchangeable Magnification, therefore it’s designed for use with multiple lenses. Aven In-X redefines what is possible with a
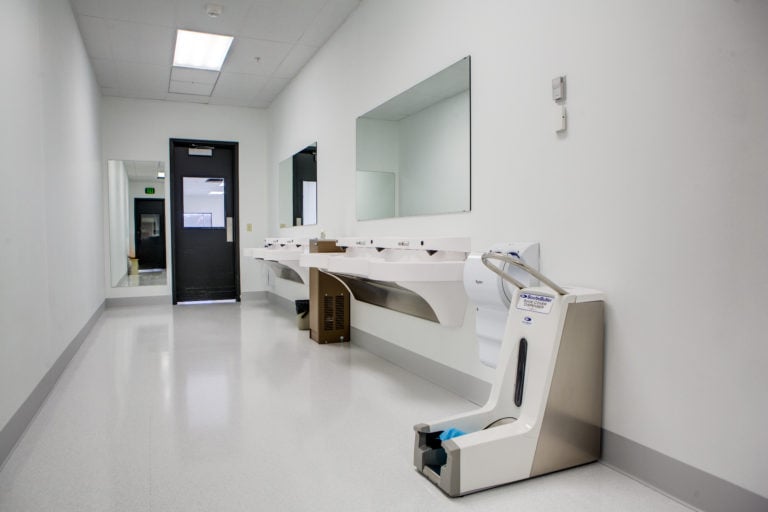
Find hand dryers, laboratory receptacles and cleanroom mirrors. New from CleanPro with quick ship options.
ISO 13485 is designed for use throughout the life cycle of a medical device. It supports each stage of medical device development and operation from initial concept to production and disposal. The standard helps internal and external parties certify and validate processes which maintain the stability of quality management systems (QMS).

Lynx EVO is an ergonomic stereo microscope without eyepieces, powering your productivity through stunning 3D viewing.

What makes MAPA gloves popular in demanding conditions? We tested the full range of MAPA gloves for form, fit and function and highlight our favorites from each category.

Select your BenchPro workbench, and save 15% on any anti-fatigue mat or Lissner chair.
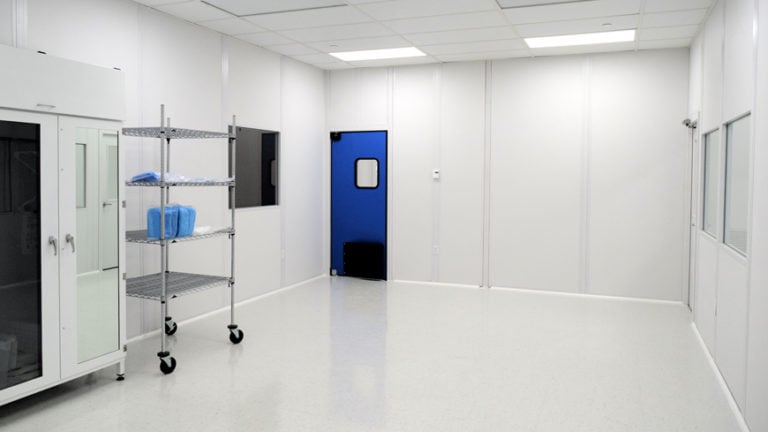
Perfex cleanroom mop and disinfection systems are designed for easy use and maintenance for critical operations. It’s the cleaning performance you’d expect beyond old-style buckets, wringers, string and sponge mops.
