Table of Contents
Surface Sanitation, Wipedown, and Disinfection
1. Follow relevant site protocol (procedures for safety, contamination, etc.) and wear cleanroom gloves.
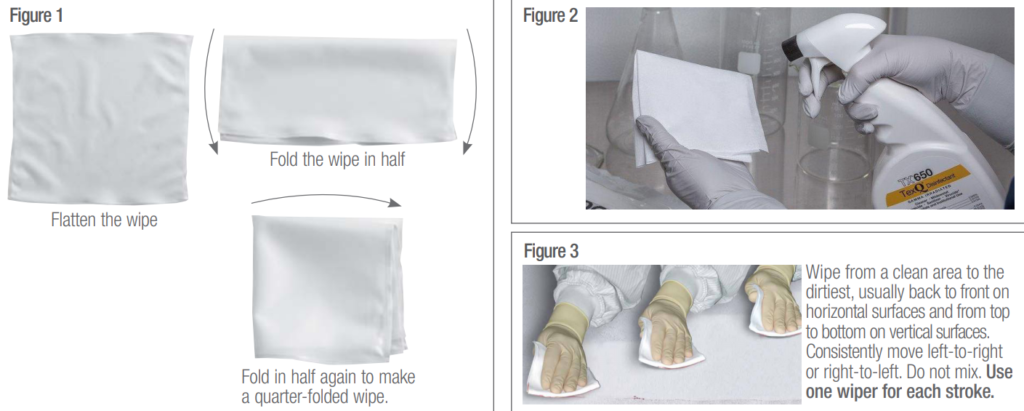
2. Fold wiper in mid-air into quarter folds (Fig. 1A). This will produce several clean surface areas and allow better contact with the surface to be wiped.
3. When wiping, hold the wiper so that the folded edge is toward the area to be wiped. Hold the unfolded edges in your hand. Group the unfolded edges between thumb and forefinger.
4. Use either a pre-wetted wiper or a dry wiper moistened with an appropriate cleaning agent. (Fig 2)
5. Wipe in one direction, overlapping wiped area by 10% to 25%.
6. Wipe from cleanest to least clean regions of the surface being wiped. Wipe systematically, for example, from top to bottom, far to near. (Fig. 3)
7. Keep track of which surfaces have been cleaned and which wiper areas are unused.
8. Always use the cleanest surfaces of the wiper. If re-wiping use a clean portion of the wiper, not the used wiper area.
9. Dispose of wipers according to site procedures.
Wiping Wet Spills
1. Identify the spilled liquid. Follow the Material Safety Data Sheet (MSDS).
2. Choose wiper and gloves that will not be degraded by the liquid.
3. For hazardous spills, wear two pairs of gloves and try to keep the gloves dry. Wear any other necessary protective gear.
4. Use dry wipers to wipe spills up immediately. Then clean the affected surfaces by following steps 1-9 in the “Cleanroom Wiping Guide” above.
5. Dispose of wipers according to site procedures.
Isolator Cleaning Guide
Preparation for Cleaning:
1. Wipe down gloved hands and cleaning products with wipers prewetted with 70% IPA and place the products in the pass-through.
2. Close the pass-through. Wait the prescribed amount of time for the pass-through to reach equilibrium.
3. Wipe gloved hands with 70% IPA; place gloved hands into the isolator gloves.
4. Open the interior door to the pass-through and bring all components into the isolator interior. Close the pass-through door. Wait for the prescribed amount of time for the isolator to reach equilibrium.
Cleaning the Biological Isolators and Laminar Flow Cabinets:
For surfaces not within arm’s reach, use the Isolator Cleaning Tool and mop cover moistened with IPA (Fig. 1).
For surfaces within arm’s reach, use a quarter folded wiper (Fig. 2) wetted with IPA (or use a pre-wetted wiper).
1. Begin by wiping the top surface (‘ceiling’) with care so as not to damage the filter, then proceed to the back wall, then the sidewalls and finish with the deck (Fig. 1).
2. Work from high to low, clean to dirty, dry to wet.
3. Wipe in straight, parallel, overlapping strokes (Figs. 1 and 2). 4. Change wiper surfaces with each stroke, and mop covers as needed.
5. Use IPA moistened swabs to clean the corners and small, tight areas.
6. Place the used, soiled wipers, swabs and mop covers in a disposal bag or place through the disposal port (if applicable).
Protocol for Hazardous Drugs Removal
Remove any excess liquid from the surface to be decontaminated using a sterile Texwipe dry wiper*. Discard the contaminated wiper in a manner consistent with the facility SOP.
- Spray the TexQ® TX650 cleaner and disinfectant on a sterile dry wiper*.
- Quarter fold a wiper. See Figure 1.
- With the nozzle on stream, not spray, pump the TexQ® TX650 trigger four or five times, until the wiper is well dampened, but not wet and certainly not dripping.
- Wipe the surface with the TexQ-wetted wiper, using parallel, unidirectional overlapping strokes. Use one wiper for each stroke (up to 40 inches in length). See Figure 3.
- Repeat Steps 2–5 using the same wiping technique at ninety degrees (90°) to the previous wiping direction.
-
EPA-registered, one-step disinfectant and cleaner
-
Effective against some Human Coronavirus, Canine Coronaviruses and Transmissible gastroenteritis, which is also a type of Coronavirus for pigs.
-
For use with 100% polyester wipes only on hard, non-porous surfaces. Cellulose and cotton fibers deactivate any Quaternary disinfectant.
TexQ® Disinfectant does not yet have a 2019-nCoV kill claim for the latest novel corona virus strain.
Sources
Cleanroom Wiping Guide (Surfaces) – Texwipe
CDC – How to Clean & Disinfect for COVID19
Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008
A Rational Approach to Disinfection and Sterilization
Basics of Biological Safety Cabinet (BSCII) Decontamination
Disclaimer
Consulting a contamination control specialist about your specific needs or requirements is best practice. The information herein is for discussion only does not constitute a recommendation for your facility.
Effective cleaning requires a case-by-case evaluation and consideration for state, federal, and local authority. It is the responsibility of the manufacturer to outline, define, revise, and validate standard procedures and protocols internally.
Web content provided by Production Automation Corporation (PAC) makes no claims as to the final interpretations or implementation of regulatory documents, organizational standards, or best practices as provided by stakeholders, CDC, FDA, WHO, OSHA, Poison Control, State Board of Pharmacy, or United States Pharmacopeia (USP).
Related Posts
-
Cleanroom Wipe Use, Protocol, and ISO Class Standards
Cleanroom wipes differ by application, cleanliness, materials, and essential properties. This post addresses cleanroom wipe ISO classes and properties. What's the right wipe for ISO class 5, ISO 6, ISO 7, or ISO 8??
-
Cleanroom Wipes for Pharmaceuticals: Pyrogen Testing Methods
Cleanroom wipes and gloves undergo LAL and Agar Overlay Analysis when testing for pyrogen and endotoxin levels. This post details the methodology, historical relevance, and explores cleanroom solutions for pyrogen-free and low-endotoxin gloves and garments.
-
New: Texwipe Cleanroom Products
Texwipe offers a variety of dry, pre-saturated, and sterile wipes to meet the strict demands of controlled environments. Texwipe swabs are manufactured to exacting and consistent tolerances with lot coded for traceability and quality control.
-
Particulate and Molecular Contamination Tests for Cleanroom Wipes and Fibers
The majority of wiper contamination tests probe for particles, fibers, ions, nonvolatile residue, and bio-contaminants. Cleanroom wipe test methods detail and measure the extraction and enumeration particles, fibers, and other contamination released from wipers (or…
-
Polycellulose & Polyester Cleanroom Wipe Supplies
Here's the guide to buying cleanroom wipes that stay on-budget. Differing package sizes, bulk counts, edge types, material options, cloth size, weights, ISO class, substrates, ionic levels…comparing cleanroom wipes is mind numbing. 50% of leading…
-
New: Perfex Cleanroom Mop & Disinfection Systems
Perfex cleanroom mop and disinfection systems are designed for easy use and maintenance for critical operations. It's the cleaning performance you'd expect beyond old-style buckets, wringers, string and sponge mops.
-
Cleanroom Wipe Testing Standards for ASTM, IEST & ISO Organizations
ISO cleanroom wipe classification is important, but is it enough? A clean room wipe selection criteria should meet baseline particulate shed thresholds for the end-user environment. Like any cleanroom rated product, cleanroom wipe manufacturers qualify…
-
Proper Material for ESD Cleanroom Gloves
ESD cleanroom glove materials have reduced NVR (non-volatile residues) levels for better electrical conductivity. Meanwhile, dedicated ESD fabrics are often not viable for cleanroom use, as they tend to shed particulates at a much higher…
-
Polycellulose & Polyester Cleanroom Wipe Supplies
Here's the guide to buying cleanroom wipes that stay on-budget. Differing package sizes, bulk counts, edge types, material options, cloth size, weights, ISO class, substrates, ionic levels…comparing cleanroom wipes is mind numbing. 50% of leading…
-
Cleanroom Wipe Contamination Profiles for Medical Device Wipedown & Electronics
The ideal cleanroom wipe must meet industrial and regulatory standards concerning chemical, physical, and biological compatibility. Wipe fabrics undergo a strict examination to simulate manufacturing outcomes in the real world. Stretch and strength, chemical compatibility,…
-
Cleanroom Wipe Testing Standards for ASTM, IEST & ISO Organizations
ISO cleanroom wipe classification is important, but is it enough? A clean room wipe selection criteria should meet baseline particulate shed thresholds for the end-user environment. Like any cleanroom rated product, cleanroom wipe manufacturers qualify…












2 thoughts on “How Do I Properly Wipe Down Surfaces in Cleanrooms and Labs?”
Mitch Walleser stands among the best with informed, responsive, helpful information and replies. And he is uncommonly kind and thoughtful to the rare but annoying goofball. Professional when we need it most. Keep up the good work, Mitch.
Thanks Charlie 🙂