What is Ethanol Extraction?
Rotary evaporators are an important tool in laboratories around the world. While this technique has many different applications, this post regards the requirements of high purity ethanol and rotary evaporators for cannabinoid and terpene extraction. When combined with advanced solvent extraction and distillation processes, raw cannabis and crude cannabis yield shatter, THCA crystals or THCA-rich oral formulations. Final packaging includes many different forms of cannabis products including oils, tinctures, vaporizable concentrates, and edibles. Generally, rotary evaporators allow one of the most efficient and environmentally friendly ways of removing a volatile solvent from a non-volatile sample. Because the solvent is recoverable and reusable, it minimizes waste and also overhead expenditure.
How Is Ethanol Used for Cannabis Extraction?
Ethanol extraction is a process in which waxes and lipids are separated for the most desirable products like terpenes, flavonoids, and cannabinoids. One reason that rotary evaporation is favorable for this technique is that the process is efficient with minimal labor. Because the ethanol can be reclaimed once extracted in a rotary evaporator, a manufacturer significantly reduces overhead costs.

Key Definitions
ExtractA substance made by extracting a part of a raw material, often by using a solvent such as ethanol or water. Extracts may be sold as tinctures, absolutes or in powder form.
ExtractionDuring this process, a solid is placed in a solvent to remove soluble components. Making coffee or tea are common examples of extraction.
DistillationIn this process, a liquid or component is isolated by selective heating, vaporization, and condensation. Distilleries use this to make spirits from a mash mixture of fermented grains.
[1] (Labx.com, 2018)
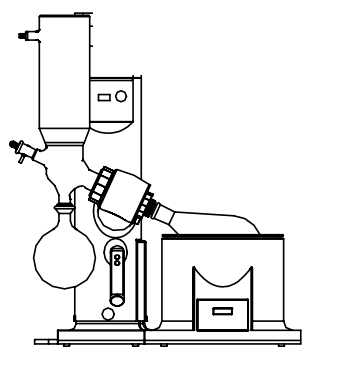
How Does a Rotary Evaporator Work?
Also known as a “rotavap”, rotary evaporators use vacuum distillation to evaporate solvent from raw or processed materials. Commonly, a cold finger condenser connects a solvent trap which sits opposite of a flask on a rotator unit. Cold water in the condenser creates a zone of cooling. The boiling flask encourages evaporation, while the solvent trap is often cooled to discourage evaporation, therefore, trapping volatile compounds once they have begun to accumulate in a liquid phase.
Rotary Evaporator Vacuum Extraction
The introduction of a vacuum allows greater distillation capacity. Boiling points and system pressure are codependent. System pressure and water temperature is adjusted to provide the ideal boiling point temperature. As the temperature of a bath or pressure lowers, the evaporating solvent could exceed the condensation capacity of the evaporator, which causes “bumping” or unnecessary solvent loss.
Rotary evaporators allow many configurations depending on the use. A diagonal glass condenser is common for standard distillation of non-volatile substances. Vertical class condensers allow solvents with higher boiling points. For ethanol extraction with dry ice, a rotary evaporator with a cold finger glass condenser is ideal. A coldfinger configuration is common for the production of tinctures, yielding a thick concentrate that is considerably pure with relatively little solvent use.
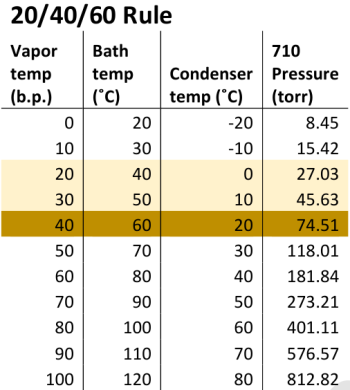
Water Bath Temperature
As a general rule of thumb, the water bath temperature is set twenty degrees higher than the desired vapor temperature. For ethanol, the recommended vapor temp is 15-20 degrees C.
Improper temperature configuration causes air within the rotary evaporator to saturate with the solvent. This is a leading cause of solvent loss. Maximizing solvent recovery requires an evaporator with a high degree of vacuum control and integrity within the air seals and gaskets throughout the vacuum line.
Vacuum Distillation
This process uses distillation under reduced pressure. Placing a liquid mixture under vacuum enables the distillation process to occur at a lower temperature. This lowers the liquid boiling point, increases the rate of distillation and reduces exposure of temperature-sensitive components (this eliminates unwanted degradation due to high heat exposure). A simple example of the difference pressure makes to the boiling point: Water at sea level boils at 212°F (100°C); in Denver, CO, it boils at 203°F (95°C) due to lower atmospheric pressure.
Ethanol ExtractionFCC grade ethanol is a superior grade solvent to extract plant material. This can be used in a variety of vessels from reactors to barrels. A popular process has the ethanol chilled to <-20°C (-4°F) either in a cold room or freezer and then pumped into a container of cannabis. After a soak period, the ethanol solution is either filtered or the plant material removed in a ‘tea bag’ fashion.
The resultant mother liquor of ethanol and extract is then concentrated by removing the ethanol. Typical distillation apparatus used to remove the ethanol include rotary evaporators or a vacuum distillation system. If a jacketed vessel or jacketed filter reactor is used to cool ethanol for the extraction process, a recirculating chiller acts as the cooling source.
[1] (Labx.com, 2018)
What is the Advantage of Rotary Evaporation for Ethanol Extraction?
Rotary evaporators extract solvents at low temperatures with a high degree of repeatability and efficiency. The method of action decreases the pressure inside of the round bottom flask and increases the surface area through gentle rotation which improves process time. Gentle agitation promotes thorough evaporation and reduces the risk of bumping or flash boiling. The term “bumping”, refers to when a large pocket of solvent vapor forms rapidly and displaces the surrounding liquid.
Ethanol Winterization Process
Winterization is the process of removing waxes and lipids to create a consumer ready product. Waxes and lipids often produce unfavorable traits when heated and vaporized. Unless removed, consumption requires more heat, diminishes flavor, increases residual residues, and may cause harshness or irritation.
After winterization, the product is dissolved in ethanol and passes through vacuum lines into an evaporation chamber and on to a rotating flask which floats over a waterbath. The flask spins in order to create a thin film on the wall of the flask, causing slow and controlled precipitation of any volatile solvents.
When exposed to heat, this experiment shows that a mild water bath is favorable over intensive heating. For ethanol extraction, dry ice within the vertical “coldfinger” silo allows ultra-low temperatures which coax a complete removal of residual solvents from the target product. Room temperature evaporation often produces greater volumes of waxes and pigments. These byproducts may produce discoloration, harshness, or less palatable flavors, hence super-chilled extractions remain preferred.
Rotary Evaporation Temperatures
A vacuum pump lowers a solvents boiling point by reducing pressure within the rotary evaporator. The water bath is deployed at 30-40 degrees with an ethanol vapor temperature 15-20 degrees C. Lower water bath temperatures prevent thermal decomposition. Increasing the evaporation rate by lowering the vacuum and/or increasing the water bath temperature can lead to capacity overload on the condenser. (Julabo)
Best Ethanol for Rotary Evaporator Extraction?
FCC grade ethyl alcohol (ethanol) is a mounting standard during concentrate extraction and food-related cannabis products. FCC approved products designate the highest purity for consumable products with no contaminants such as denaturants, heavy metals, methanol, hexane, and benzene. The purest solvents allow perfectibility and more consistent yields while also producing cleaner, better tasting concentrates.
Ethanol Flammability
Ethanol meets the criteria for a Category 2 flammable liquid, according to OSHA’s Flammable Liquids standard and the HCS (29 CFR 1910.106; 29 CFR 1910.1200 Appendix B). This is because ethanol ignites at normal room temperatures, has a flash point of 55°F, and has a boiling point of 173°F [2] (OSHA/EPA, 2011).
Rotary Evaporator Configuration Diagrams & Images
The method of action decreases the pressure inside of the round bottom flask and increases the surface area through gentle rotation which improves process time. Gentle agitation promotes through evaporation and reduces the risk of bumping or flash boiling.
The water condenser is replaced by a cold trap that enables very low freezing temperatures. Hence, with this assembly, it is also possible to completely condense extremely volatile and low-boiling solvents such as chloroform or dichloromethane. You can fill the cold trap not only with common freezing mixtures such as dry ice/acetone (-70°C) or ice/common salt (-20°C) but also with liquid nitrogen (approx. -190°C). This allows you to perform sublimations as well. The cold trap does not require running water for cooling; therefore, it saves water.
Sources
- Labx.com. (2018). The Cannabis Workflow and the IMPORTANCE OF TEMPERATURE CONTROL. [online] Available at: https://www.labx.com/resources/the-cannabis-workflow-and-the-importance-of-temperature-control/71 [Accessed 21 Dec. 2018].
- Osha.gov. (2018). OSHA Technical Manual (OTM) | Section IV: Chapter 5 – Ethanol Processing | Occupational Safety and Health Administration. [online] Available at: https://www.osha.gov/dts/osta/otm/otm_iv/otm_iv_5.html [Accessed 21 Dec. 2018].
- Hoegger, R. (2018). Distillation with a Rotary Evaporator. [online] Imlab.be. Available at: http://www.imlab.be/imlab_nl/buchi/distillation_rotary_evaporator_imlab.pdf [Accessed 28 Dec. 2018].3
- Nwsci.com. (n.d.). Rotary Evaporators. [online] Available at: https://www.nwsci.com/customer/docs/SKUDocs/RMR/Rotary%20Evaporator.pdf [Accessed 2 Jan. 2019].
Related Posts
-
Ethanol Purity, Grades, and Extraction Techniques
This post covers the unique characteristics of high purity ethanol for commercial cannabis extraction. We’ll outline methods that improve batch efficiency, reduce terpene loss from decarboxylation, and minimize residual solvents. In large-scale extraction environments, closed-loop…
-
Why Do You Need Fume Extraction?
In today's industrial and service environments, maintaining clean air is not merely a regulatory requirement but a vital necessity. Exposure to harmful fumes and particles can lead to severe, long-term health issues, adversely affecting worker…
-
New: Elma Ultrasonic Cleaners
Elma Ultrasonic Cleaners - Where to Buy Online? Elma introduced its ultrasonic cleaners to the world over a half century ago. Today, its systems stand among the world's best.Ultrasonic cleaners excel for rapid, intensive cleaning…
-
Branson Ultrasonic Cleaners
CSA-listed cleaners provide thorough, safe cleaning of small- and medium-sized parts Seven sizes give you the choice of just the right capacity cleaner for your needs Recessed cleaning tank minimizes spills and drips Specialized models…
-
BOFA Fume Extractors for 3D Printing
3D printers are known to produce a number of byproducts during operation. Common 3D printing filaments such as ABS and PLA are known as high emitters of ultra-fine particulates (UFPs), while nylon and PCTPE are…












1 thought on “Rotary Evaporators for Ethanol Extraction”
I am confused. You show a photo of a diagonal condenser for ethanol extraction at the top of the article, then say diagonal condensers are not good for volatile chemicals. Will the diagonal condenser work for hemp oil extraction using ethanol?