
Cleanroom Fan Filter Replacement
When Should I Replace a Cleanroom Fan Filter? Pre-Filter Replacement Pre-filter media should be replaced every six months and inspected regularly. Pre-filters in gowning rooms

When Should I Replace a Cleanroom Fan Filter? Pre-Filter Replacement Pre-filter media should be replaced every six months and inspected regularly. Pre-filters in gowning rooms

A cleanroom air handling unit (AHU) is a cornerstone of temperature, humidity, and pressurization within controlled environments. Today, modular fan filter units allow more adaptable configuration with simplified maintenance procedure. Both systems serve conditioned and filtered air throughout a cleanroom facility.
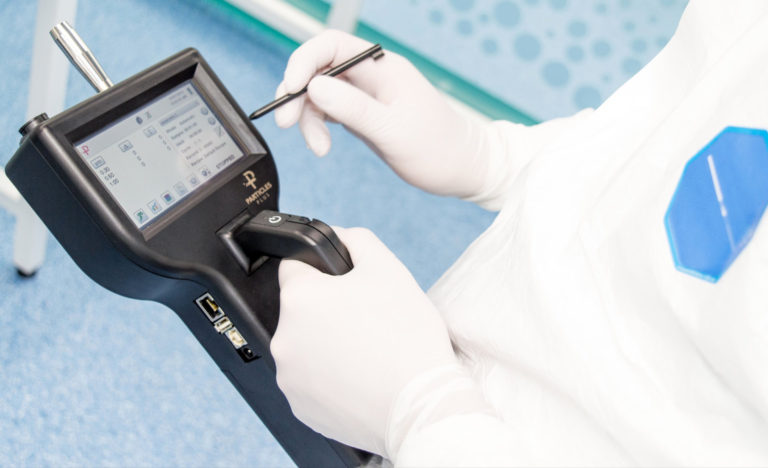
For ISO 14644 compliance, all particle counting equipment requires a calibration certificate.

Updated: USP 800 is a cleanroom standard issued in March of 2014 by the United States Pharmacopeial Convention (USP). The deadline December 2019 for compliance may change.

Pre-engineered fabrication consolidates scheduling with minimal disruption and fewer variables. Difficult or expensive upgrades avoid overbearing noise, construction artifacts, and minimize if not eliminate the burden on facility staff for project management.

USP Chapter 797 cleanroom design requires that facilities pressurize non-hazardous compounding and storage areas. ISO 5, 7 and 8 environments support primary engineering controls, buffer rooms, and ante-rooms. Updated: 4/18/2019

New USP guidelines may present challenges for compounding facilities. Some facilities need infrastructural and mechanical modifications for compliance. System evaluation includes duct systems, HEPA fan filters, differential pressure standards, air monitoring, and external air exhaust equipment.
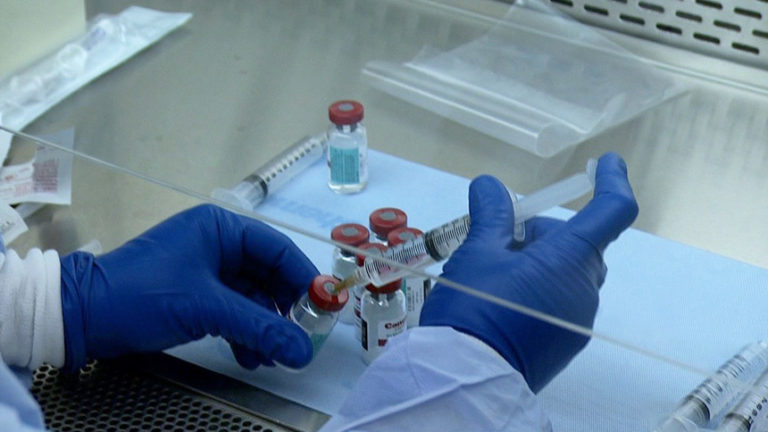
Facilities unpacking hazardous drugs within a negative pressure room do not require procedural changes to meet USP unpacking area requirements. Facilities unpacking HDs within positive pressure ante rooms or facility spaces will require reconsideration.

The definitive cleanroom and sterile compounding glossary. Learn terminology and definitions for USP 797 and USP 800 compounding.

How does a cleanroom adapt for a medical cannabis grow? It starts with specialized containment rooms, HVAC, air showers, clean surfaces, and contained lighting systems. Are cleanrooms an essential piece of achieving high quality, high yield medicinal products for licensed cannabis growers?

The benefit of modular cannabis cleanrooms over traditionally fixed wall grow rooms is that the configurable partitions of modular cleanroom designs lower the cost of installation or re-configuration for changing regulations. Divider rooms total environmental control, larger yields, and improve regulatory compliance at various stages of the growing cycle.

Cleanroom HEPA Vacuum Components and Applications Simply put, every square inch of a cleanroom should be absolutely pristine. Ceiling panels, lighting units, HEPA filtration units,

What are the key determinants of cleanroom components? Learn about different types of cleanroom filtration, wall construction, lights, temperature, humidity and more.

Proper cleanroom cleaning procedure and maintenance protocol is an immediate, low-cost measure to enhance overall cleanliness, consistency, and contamination control within manufacturing and aseptic cleanrooms. This guide provides a framework for cleanroom management, protocol standards, specifications, and processes for general manufacturing and bio-medical applications.

Advantages for Modular Hardwall Design and Construction of ISO 14644-4 Cleanrooms.

High-traffic cleanrooms use tightly controlled procedures with repeatable processes to maximize the passage of people in and out of sanitary environments. Cleanroom facilities of all

There are numerous considerations when evaluating cleanroom construction and design. The goal of this post is to touch on the basics. You may very well

1. Ignoring the Need to Wipe on a Regular Basis To assure quality is maintained in all cleanroom and restricted areas, housekeeping maintenance must be

Article reprinted from Clean Air Products, manufacturers of air showers, cleanrooms, and cleanroom accessories. A cleanroom provides a controlled environment in which companies can produce

Palbam Class Stainless Steel Cleanroom Desiccator Cabinets Palbam Class developed its first desiccator cabinets for the Semiconductor Industry. Due to the demands of this industry
