
GMP Cleanroom Design Standards Part 1: GMP and CGMP Manufacturing
What is GMP? What is CGMP? What are the differences between GMP and CGMP cleanrooms?

What is GMP? What is CGMP? What are the differences between GMP and CGMP cleanrooms?

Cleanroom wipes and gloves undergo LAL and Agar Overlay Analysis when testing for pyrogen and endotoxin levels. This post details the methodology, historical relevance, and explores cleanroom solutions for pyrogen-free and low-endotoxin gloves and garments.

ISO cleanroom wipe classification is important, but is it enough? A clean room wipe selection criteria should meet baseline particulate shed thresholds for the end-user environment. Like any cleanroom rated product, cleanroom wipe manufacturers qualify ISO wipers between ISO Class 1 – ISO Class 9 as specified in step with cleanroom classifications of the International Standards Organization (ISO). The majority of cleanroom classifications fall between ISO 5 – ISO 8.

What are the steps for medical device cleanroom validation? Cleanroom validation ensures cleanroom construction meets the performance standards set forth during cleanroom planning, cleanroom installation, and cleanroom calibration.

How do I know when to replace my cleanroom HEPA air filters? How do I test a cleanroom HEPA filter? What are the best practices for cleanroom HEPA filter replacement?

The majority of contamination within aseptic processing cleanrooms involves personnel. Proper application of gowns, hygiene, and a work flow can often eliminate the majority of mix ups and contamination.

When Should I Replace a Cleanroom Fan Filter? Pre-Filter Replacement Pre-filter media should be replaced every six months and inspected regularly. Pre-filters in gowning rooms

Warm water and a mild dish soap is the best softwall curtain cleaning solution. What about acrylic, melamine, and aluminum? Generally, any biodegradable solution is acceptable. A 50/50 solution of Isopropyl Alcohol & DI water is also effective. Wash curtains front side and back, rinse with clean water, then wipe dry with a clean lint-free soft cloth.

First published in 2004, USP Chapter <797> has undergone proposed revisions as of July 2018. These revisions are now available for public comment.
ISO 13485 is designed for use throughout the life cycle of a medical device. It supports each stage of medical device development and operation from initial concept to production and disposal. The standard helps internal and external parties certify and validate processes which maintain the stability of quality management systems (QMS).
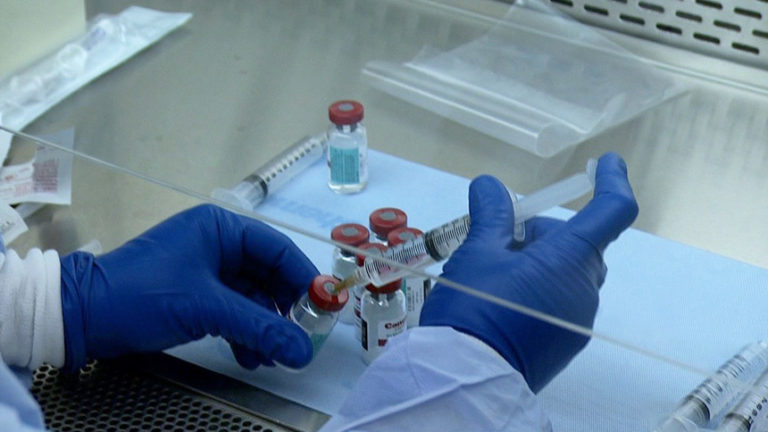
Facilities unpacking hazardous drugs within a negative pressure room do not require procedural changes to meet USP unpacking area requirements. Facilities unpacking HDs within positive pressure ante rooms or facility spaces will require reconsideration.

Cleanroom HEPA Vacuum Components and Applications Simply put, every square inch of a cleanroom should be absolutely pristine. Ceiling panels, lighting units, HEPA filtration units,

This guide provides a framework for employees entering a cleanroom. The thousands of people who all wear “bunny suits” to protect products from from human particles such as skin flakes or hairs. A bunny suit is made of unique, non-linting, anti-static fabric and is worn over street clothes.

Proper cleanroom cleaning procedure and maintenance protocol is an immediate, low-cost measure to enhance overall cleanliness, consistency, and contamination control within manufacturing and aseptic cleanrooms. This guide provides a framework for cleanroom management, protocol standards, specifications, and processes for general manufacturing and bio-medical applications.

Learn how to properly mop a cleanroom for ISO class or cGMP cleanliness levels. Compare designs and materials of multi-bucket cleanroom mop systems.
Cleanroom wipes differ by application, cleanliness, materials, and essential properties. This post addresses cleanroom wipe ISO classes and properties. What’s the right wipe for ISO class 5, ISO 6, ISO 7, or ISO 8??

How to Certify a Cleanroom with Kanomax Hand Held Particle Counters

High-traffic cleanrooms use tightly controlled procedures with repeatable processes to maximize the passage of people in and out of sanitary environments. Cleanroom facilities of all

1. Ignoring the Need to Wipe on a Regular Basis To assure quality is maintained in all cleanroom and restricted areas, housekeeping maintenance must be

Read the complete guide to decontamination and disinfection of biological safety hoods and cleanroom isolators for pharmaceutical production and laboratories.
