Table of Contents
The Guide to Low Lint Cleanroom Wipes for Cleanroom Cleaning & Maintenance
Cleanroom wipes differ by application, cleanliness, materials, and essential properties. This post addresses cleanroom and industrial requirements such as ISO classes, procedures, protocol, ESD-safe properties and use cases for manufacturing. The cleanroom wipe is historically the most effective and commonly used tool for the removal of contaminants, particulate, and residual material in critical manufacturing and pharmaceutical compounding environments. Highly absorbent, low lint materials enable tight control of moisture, solvents, and spills for cleaning of cleanroom surfaces, walls, machinery, fixtures, transport carts, tools, and other production materials.
What’s the Difference Between a Woven and Non-Woven Cleanroom Wipe?
What's a Nonwoven Wipe?
Nonwoven fabrics are broadly defined as a sheet or web structure bonded together by entangling fiber or filaments (or by perforating films) mechanically, thermally or chemically. Post-treatment and unique construction characteristics provide some potentially beneficial outcomes, such as a porosity that enables gas sterilization. Nonwoven wipes offer an economical choice, as the manufacturing process does not require laundering. Cellulose and synthetic blended hybrids provide additional absorbency for spill cleanup and removal of thick liquids. Generally, nonwoven wipes are used in cleanrooms no cleaner than ISO Class 6 or above.
Made with a hydroentangled blend of 45% polyester / 55% cellulose; providing economical performance & purity. This creates a strong, clean, and very absorbent wipe for liquid removal or lint-free wiping in controlled areas.

What's a Knit (Woven) Cleanroom Wipe?
Woven wipes, also known as knitted wipes, assist contamination control in critical environments. Woven fabrics are the highest quality cleanroom wipes on the market today. Low levels of NVRs and superb particle removal is ideal for critical class environments. Unlike nonwoven fabrics, woven cleanroom fabrics are formed by knitting or looping monofilament, continuous polyesters or other synthetics. The outcome is cleanroom wiper with great tensile strength that will not shed. The lowest amount of linting and particulate generation yields compliance for ISO Class 3 and upwards.
What Are the Advantages of Woven Polyester Cleanroom Wipes?
100% Continuous Filament Polyester — What is a Knitted Polyester Cleanroom Wipe?
Polyester is a synthetic petroleum-based fiber. Monofilament or continuous polyester cleanroom wipers is the cleanest wiping material available. Monofilament structure ensures a very strong and durable material. Naturally, polyester absorbs solvents but not aqueous solutions; a finish treatment is commonly applied to improve sorbent properties.
Laundered polyester knit cleanroom wipes undergo a laundering process using ultra-filtered water at 0.45 micron before HEPA filtered moisture removal. Packaging and sealing under an ISO class 4 laminar flow workstation with a hermetically sealed (air tight) package ensures cleanliness and traceability before final inspection. Cleanroom processing and laundering ensures low particle shed, consistent quality, and favorable outcomes in class 3-5 cleanrooms. The wipe will also not shrink or change shape. Compatibility with cleaning solvents such as IPA and autoclave ready temperature ratings enables use for the most critical cleanroom environments.
Double Knitted Polyester Cleanroom Wipes
Double-knitted monofilament polyester creates a super soft fabric for increased durability and particle retention. The material is knitted and interlaced twice in a cross pattern for the strongest possible outcome. Double knits yield a cross-hatched texture which increase surface area and capture more particles than traditional polyester knit wipes. Tightly knitted designs reduce surface catches, unraveling, and breakage of yarn filaments that generate particulate.
Polyester Double Knit Wipes The CleanPro® Double Knit Pro 1 9″ x 9″ wipe is composed of 100% double-knit polyester. They are processed and double bagged in an ISO Class 4 cleanroom environment.
Polyester/Nylon Cleanroom Wipes
Polyester/nylon wipes for cleanroom and industrial applications generally feature a mix of between 25-30% nylon and 70-75% polyester. The result is a very soft fabric which will not scratch sensitive devices yet is extremely effective in trapping micro-contaminants without re-contaminating the surface.
Polyester/Cellulose or Polycellulose Cleanroom Wipes
Polyester cellulose wipes, also known as polycellulose wipes, generally feature a 55/45 mix of cellulose and polyester which is hydroentangled for a chemically pure wipedown without binders, starches, or thermal bonding. Cellulose aids electrostatic dissipation as a naturally ESD-forward material, which is commonly used where liquid retention is important.
What are the Advantages of Cellulose Blended Wipes?
The mix of cellulose and polyester may vary as the majority depending on the need of absorbance versus less lint. Cellulose is more absorbent, while polyester is less linting. Polycellulose blended wipes are one of the most popular cleanroom wipes for general wipedown and spill control in ISO Class 6 and above cleanrooms. Biodegradable and naturally ESD friendly, polycellulose provides one of the most economical cleanroom wiper options because it’s easy to produce and process. Price, chemical resistance, and high absorbency are among its most desired features.
Types of Nonwoven Cleanroom Wipes
Nonwoven fabrics are flat, porous sheet made from separate fibers or from molten plastic. Conversion from fiber to yarn for weaving or knitting is unnecessary in the manufacturing process. Nonwovens have a limited lifespan, and are generally used for one time wipe down and then discarded. Non-woven materials support a number of applications where a unique combination of characteristics is achieved by a combination of fiber and additives. An advantage of nonwoven fabrics is the capability for very wide widths, and an overall lower cost of manufacturing. Three types of manufacturing processes distinguish most nonwoven cleanroom wipe designs:
Meltblown wipe construction requires the thermal bonding of ultra-fine polypropylene fibers.
Hydroentangled wipe construction requires high pressure jets which entangle and clean the fibers. The process forgoes binders or additives which yields the lowest possible residual remnants or manufacturing bi-products like binders, ions, or extraneous fibers.
Spunlaced wiper construction applies low impact water jet energy to a fabric web and vacuum dewatering that results in spunlaced fabric. Low particle counts when wet or dry renders spunlaced wipers particularly useful for cleanroom wipe down.
Cotton Cleanroom Wipes
Cotton provides a thermally stable, solvent resistant, and static dissipative wiper within an ISO Class 7 or above cleanrooms or clean spaces. Its ability to absorb up to 6x as much liquid as other wipes is ideal for handling aqueous or solvent solutions. While cotton is not recommended for critical particle control, it enables high heat cleaning for enclosed injection molding, electronics, or other enclosed high temperature machinery. Cotton enables elevated temperature applications, and food-safe contact due to natural construction which is biodegradable and sustainably sourced. The most delicate of common cleaning fabrics, cotton is uncommon where minimal shedding and tear resistance is paramount. Despite claims of cotton wipes that attain ISO Class 3 – ISO Class 5 compliance, our in-house experts agree that a cotton wipe should not be used in an ISO Class 6 cleanroom or cleaner.
Polypropylene Cleanroom Wipes
Polypropylene is an industrial-grade thermoplastic which when combined with minerals and other additives during the manufacturing process is malleable into synthetic papers and wipes. The result of meltblown entanglement process is a uniformly flat surface with exceptional particle removal characteristics. It has excellent compatibility with acids, bases, and solvents.
Microdenier or Microfiber Wipes for Cleanroom Applications?
Microdenier, interchangeably used with microfiber, is constructed of synthetic yarn, typically a bi-component polyester-nylon polymer, or 100% polyester polymer. (View PDF) Microfibers absorb up to 99.4% of microorganisms found on the surface compared to only 30% for wipers containing cellulose. Therefore, microfiber is ideal for cleaning nano-sized particles, removing fingerprints from screen displays, applying or removing liquid, or wiping a wet surface dry. Problematically, the intense absorption characteristics render a material that is difficult to clean, and also retains many of the additives, ions, particulate and NVR acquired during the manufacturing process. Fragile bicomponent filament construction, such as 70% polyester and 30% nylon challenges particle control because it’s more susceptible to tearing, breaking, and shedding. For cleanroom use, a 100% polyester microfiber yarn ensures a cleaner more consistent wipe due to tightly knit filament structure which exceeds the cohesiveness of the previous generation of microfiber materials.
Dry and Presaturated Cleanroom Wiper Materials and Construction
Presaturated Cleanroom Wipes
Local or large-surface disinfection of cleanrooms or clean spaces favors easy to grab, easy to apply, and evenly saturated solvent wipes. Simplified workstation and equipment wipedown provides a high level safety and procedural consistency. Presaturated wipes aid cleanliness in compounding pharmacies, pharmaceutical manufacturers, and hospitals because of the convenience and even dispersion of chemical solvents.
Use of Sterile Presaturated Cleanroom Wipes
Sterile presaturated wipes generally feature a meltblown polypropylene or knitted polyester fabric saturated with 70% IPA and 30% deionized water which is then gamma irradiated. Gamma irradiation and aseptic filtration ensure sterile outcomes during the manufacturing of sterile presaturated wipes. Sterile wipedown materials ensure the utmost cleanliness in critical drug compounding areas such as intravenous IV preparation or sterile packaging. Resealable pouches and tamper-evident labels ensure integrity of the product upon final delivery.
Sterile Cleanroom Wipes
Pharmaceutical, Compounding or Medical Device Packaging
USP 797 and USP 800 compliant pharmacies, cGMP pharmaceutical compounding facilities, and medical device manufacturers require gamma irradiated cloths to maintain an aseptic environment. Sterile classification requires that biomedical and pharmaceutical products abstain from the presence of living organisms and also pyrogens. A pyrogen is a lipopolysaccharide from the cell wall of a gram-negative bacteria, which commonly grows in ultra-pure water. USP chapter 161 denotes acceptable limit of endotoxins or pyrogens with 0.5EU/mL for medical devices and 20 EU/device for products in direct contact with cardiovascular or lymphatic systems. The FDA specifies a limit of <1EU/wipe, 0.5 EU/mL for medical devices, or 20 EU/device for products that directly or indirectly contact the cardiovascular and lymphatic systems. Final delivery features triple or double-bagged packaging and a certificate of sterility.
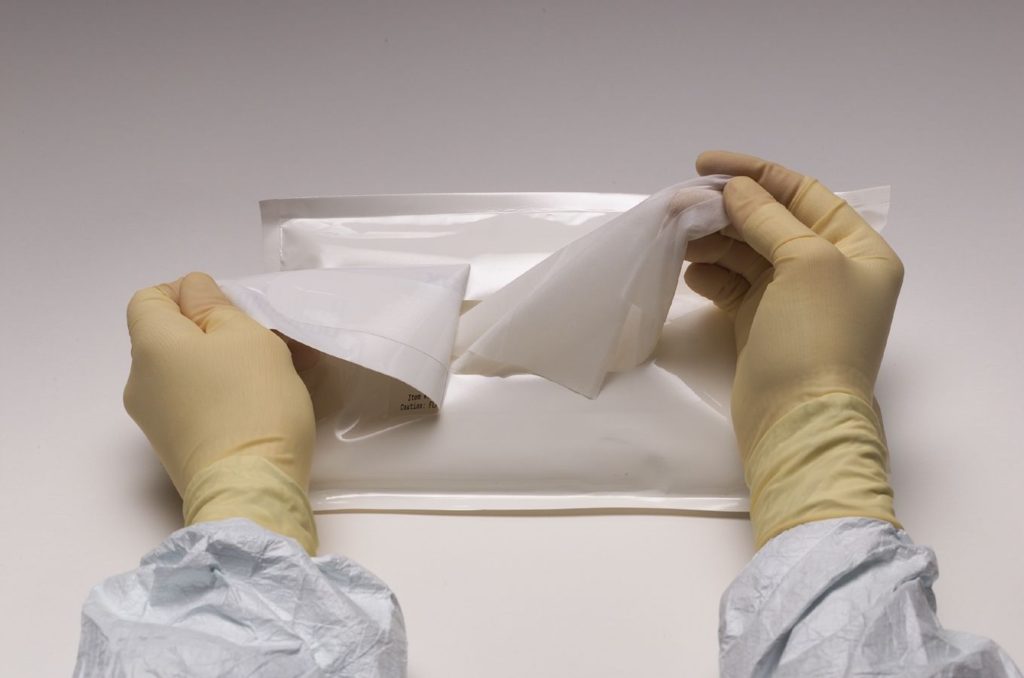
Presaturated wipes are pre-wetted and saturated with 70% isopropyl alcohol (IPA) and deionized water (DI) filtered to 0.2 um. Each pouch, usually sold by the case with individual packs of 50 wipes, is labeled with a lot number for traceability and process accountability.
Sterile IPA Cleanroom Swab Usage
70% IPA swabs are commercially available in individual foil-sealed packages. Application of IPA to entry points on bags and vials before opening curbs any preexisting contamination obtained during the transportation from its sterile packaging center. Sterile packaging designed for sterile delivery and opening does not require disinfection on site when used properly. IPA wetted gauze or other particle-generating material is not effective in this process, due to potential introduction of physical particulate within CSPs.
Cleanroom wipes and swabs halt particle shed and contaminate spread during the wipedown process of ISO class cleanrooms. The use of cleanroom wipes increases with output and degrees of cleanliness with frequency define by multiple factors, including: The beginning of a production shift, before each segment of the manufacturing or production process, after spills, or when surface contamination is known or suspected. For pharmaceutical compounding, wipedown is conducted at intervals of no longer than 30 minutes while activities remain ongoing.
The Case For Cleanroom Wiper Dispensers
Wipe dispensers provide easy storage, easy access, and eliminate the waste and cross-contamination of holding too many wipes at a single time, therefore throughput is easily scaled. Pre-saturated 70% isopropyl wipes generate ideal outcomes during sanitary wipedown. It should be noted that isopropyl alcohol does not kill bacteria or fungal spores, which you can read about in further depth here.
ISO Cleanroom Wipe Application - Compliance for Wipers
| Example Application | Cleanroom Classification | Suggested Materials | Suggested Edge |
| Semiconductor, Isolator | Class 10 / ISO Class 4 | Polyester Knits | Sealed Edge |
| Semiconductor, Aseptic Filling Area | Class 100 / ISO Class 5 | Synthetic Knits | Sealed Edge |
| Pharmaceutical Prep Area | Class 1,000 / ISO Class 6 | Polycellulose Blends | Cut Edge |
| Medical Device Manufacturing | Class 10,000 / ISO Class 7 | Cotton, Foam, or Composites | Cut Edge |
Ultrasonically sealed polyester knits (woven) cleanroom wipes are the most widely used wipes in ISO 3 cleanrooms and above, both sterile and non-sterile. Mini environments for pharmaceutical compounding or semiconductor manufacturing require an ultra-durable wipe with minimal residues or surfactants. An ultrasonic seal is more resistant to abrasion and particle shedding during use in aseptic manufacturing facilities which reduces residuals when cleaning surfaces, tools, and equipment. Specialized double knit pattern weaves enhance the entrapment and particulate removal. Multiple layers also increase sorbency.
Ultrasonic seals prevent carbon deposits that occur during the laser sealing process. Critical operations require ultra-low particulate counts. Dry knitted woven polyester cleanroom wipes provide the cleanest classification necessary for ISO Class 3 cleanrooms.
ISO Class 4 (Fed. Class 10) — ISO Class 5 (Fed. Class 100) Cleanroom Wipes
Polyester/nylon knit synthetics provide an ideal choice for an ISO Class 4 or above cleanroom. Low in particles and extractables, they are processed, laundered, and double bagged in an ISO Class 4 cleanroom environment. Laser sealed edges provide effective control of particulate with a non-abrasive edge that cleans surfaces without scratching.
A low-lint nonwoven wipe, polycellulose blend, or polyester blend wipe minimizes particle generation and levels of NVR (non-volatile residue). Less critical applications don’t require the same 100% polyester blends as more critical applications.
ISO Class 7 (Fed. Class 10,000) and Above Wipes Standards and Classifications
Cotton, foam, and composite wipes provide a heavy weight material with extraordinary tensile strength and absorbency.
Types of Cleanroom Wipe Edges
Knife cut edges provide a very soft edge which is also non-abrasive. The cutting process, generally conducted with a steel blade, generates the most extraneous fibers or loose ends for cleanroom wipe cuts. This is the most economical option for less critical cleanroom application but also the most prone to linting or particle shed.

Laser sealed edges provide extremely precise edges with minimal disruption of surrounding material. The process ensures zero loose particles or split end for ultra-clean, heavy duty cleaning. The laser process does entail trace amounts of carbon deposit.

Ultrasonically sealed wipes impart an ultrasonic heat process which melds fibers without the need for cutting by knife or the carbon deposit of laser cuts. The result is a cleanroom wipe with lower carbon levels, and a precisely formed, stable edge. Ultrasonically sealed wipes provide the highest grade of particle retention and the most cleanliness.

Cleanroom Wipe NVRs
Controlled cleanroom environments need to restrict or eliminate particulate entering a cleanroom, but also any particulate generated within it. The result is a safer production environment for electronics, pharmaceuticals, PCBs, aerospace equipment, and more. To meet requirements of a “cleanroom” as defined by Federal Standard 209E and newer ISO Standards, all cleanrooms must not exceed a particulate count as specified in the air cleanliness class.
- Fabric substrate
- Microstructure
- Sorption capacity and rate
- Particulate burden and size of surface area
- Bioburden
- Ions
- Metals
- Non-volatile residues
Cleanroom wipers contain a variety of non-volatile residues (NVR) which include surfactants, sizing compounds, waxes, oils, adhesives, electrostatic agents, and greases. Device sensitivity drives demand for wipes with less NVR and better residual absorption. In any case, a surface can never become cleaner than the wiper used. For each cleanroom wipe application or industry, different construction and material properties distinguish ideal use.
Types of Cleanroom Wipe Material
What are the advantages of dry versus presaturated IPA cleanroom wipes?
The benefit of pre-wetted wipes is one of both context and convenience. The major advantage of pre-wetted cleanroom wipes is that they minimize time costs, require minimal space and provide simple access, and eliminate the risk of a wipe with too much or not enough cleaning or solvent saturation. The location of wipes in an easy access and close-to-use location increases the likeliness of consistent procedural protocol. Presaturated wipes provide tight control of organic cleaning compound concentration and application, minimizing the number of volatile organic compounds (VOCs) spread through the air by a spray bottle.
Advantages of Dry Wipes
Because the cost of a presaturated wipe includes the cleaner and the solvent, the cost per consumable item is often more significant than a dry wipe of the same features and classification. Cleaning solvents like isopropyl alcohol present an economical option when bought in bulk; the price savings increases with volume and therefore supply scales readily to production volume.
Use Of Cleaning/Disinfection Agents During Cleanroom Wipedown
USP 797 Compliance for Wipes and Wipedown
USP regulations apply to health care institutions, pharmacies, physicians practice facilities, and other facilities in which sterile materials are prepared, stored, and dispensed. The cleanliness of compounding areas requires scrupulous attention to minimize any sources of microbial contamination (non-sterility), excessive bacterial endotoxins, variability of ingredient strength, ingredients of inappropriate quality in compounded sterile preparations (CSPs), and unintended chemical or physical contaminants. Excessive bacterial endotoxins are potentially the most hazardous to patients when administered into the central nervous system with extremely dangerous and often fatal consequences. This is demonstrated in the case of Barry Cadden, a pharmacist convicted of killing 76 people and sickening over 700 after distributing expired and non-sterile drugs which resulted in the deadliest fungal meningitis outbreak in US history.
USP 797 Wipedown Best Practices and Compliance
USP 797 compliance requires both initial and continued application of isopropyl alcohol (IPA) throughout a cleanroom session. All disinfectant, either sprayed or wiped, must be allowed to dry with respect to the manufacturer’s directions or published data for minimum contact time. Generally, IPA (70% isopropyl alcohol) remains on surfaces to be disinfected for at least 30 seconds before CSP packaging resumes.
- USP 797 requires the use of sterile 70% IPA for the disinfection of surfaces, compounding sites, and gloves within pharmaceutical applications.
- Wherever nonsterile surfaces are touched, routine application of IPA regulates any potential introduction of contaminants.
- Supplies and equipment removed from shipping carts require sterile IPA or another suitable disinfectant from a spray bottle or presaturated wipe
- All worksurfaces in ISO 7 buffer areas and ISO 8 ante rooms require daily cleaning and disinfection
- All supplies required for operations, generally before being brought into the buffer area
- Packaged compounding supplies and components, such as needles, syringes, tubing sets, and small- and large-volume parenterals, should be de-cartoned and wiped down with a disinfectant that does not leave a residue (e.g., sterile 70% IPA). Ante-areas require ISO Class 8 air quality before safe passage into the buffer areas.
- Cleaning and disinfecting surfaces in the LAFWs, BSCs, CAIs, and CACIs shall be cleaned and disinfected frequently including at the beginning of each work shift, before each batch preparation is started, every 30 minutes during continuous compounding periods of individual CSPs, when there are spills, and when surface contamination is known or suspected from procedural breaches.
- Each month, all buffer area totes and storage shelves by removing contents and using a germicidal detergent soaked lint free wipe, cleans the inside surfaces of the tote and then the entire exterior surfaces of the tote. Allows totes to dry. Prior to replacing contents into tote, wipes tote with sterile 70% IPA to remove disinfectant residue. Uses new wipe as needed.
- Wipes carts with sterile 70% IPA wetted lint-free wipe to remove any disinfectant residue. Uses new wipe as needed.
General Best Practices for Cleanroom Wiping
- Use only cleanroom approved wipers
- The wipers must be approved for the class of cleanroom being cleaned.
- Do not allow any tool to rest on the surface of a bench or table, as it should be placed on a cleanroom wipe.
- All non-packaged items entering a gowning room should be first wiped down with 70% isopropyl alcohol, or the appropriate cleaning solution.
- Once in the cleanroom, at either your work bench or immediately inside the cleanroom, complete an ultra-cleaning on your transferred items on the cleanroom cart using 30% Isopropyl alcohol (IPA) wipes. – unlv.edu
- Before putting on your safety glasses, make sure to wipe them down with an IPA wipe provided in the gowning room.
- This will ensure that the glasses are free of any particles and clear.
- Any work or tools dropped on the floor shall be considered contaminated, and must be cleaned with an IPA wipe
- Solvent-soaked wipes should always be disposed of in the proper solvent cans
- All wipes should be disposed at the end of the session
Spray Bottles Use In Cleanrooms
Cleanroom presaturated wipes avoid the pitfalls of a solution placed within a spray bottle. Exposure to light, air, and any contaminants introduced during the IPA refill process creates risks for the immediate spread of dormant fungal or bacterial spores. Because IPA is not an effective bactericidal or fungicidal cleaner, a bottle provides opportunities for lapses in procedure and widespread contamination.
The mechanism of an IPA bottle expels a liquid, and then re-pressurizes itself with air from the environment. If the outside environment is contaminated with mold or bacteria spores, the entire bottle of IPA may become contaminated and thereafter spread over the entire cleaning surface.
The reduction of contamination risks is defined by written protocol procedures. These processes define limits of storage duration, create documented tracking procedures, and evaluate the risk of potential contamination over time.
Is The Contamination of IPA Spray Bottles In Cleanrooms A Myth Or Fact?
In theory, the potential for environmental air re-aspirating through a spray bottle nozzle is plausible. Despite suspicion that bacterial spores or fungal spores could contaminate a cleanroom spray bottle, under test conditions this hypothesis is unconfirmed.
The mechanism by which a spray bottle re-pressurizes requires the exchange of external air. After expelling liquid leads to skepticism about spray bottle use in critical environments for quite some time. Scientifically, this concept is quite testable. In this controlled study, the risk of internal contamination is shown to be minimal. 45 individual USP-grade 70% sterile IPA solutions were distributed within three environments of different cleanliness. 15 of the bottles were maintained in an unregulated warehouse. Twice each day, over a period of 30 days, the bottles were triggered. The nozzles were left open for the duration of the study. At intervals of 14, 21, and 30 days, 4 – 5 bottles were combined, sampled and collected. After testing at an independent library, all the samples indicated sterile outcomes.
In conclusion, these studies are relatively limited, but indicate that extrinsic contamination of an IPA spray bottle is difficult to repeat even in the least controlled of environments. In controlled environments, the tight maintenance of sanitation and disinfection scuttle favorable environments for fungal growth and residual spores. Notably, the results of these studies remain speculative, and demonstrate a small sample size. Further, the likeness that an area is heavily contaminated with fungal spores and that an IPA bottle would be contaminated after one spray each day is unlikely. In any case, the utmost precaution requires a procedural plan which regulates the labeling, use, and eventual disposal of all cleaners and solvents on a trackable and consistent basis.
Related Post: The Seven Sins of Cleanroom Wipedown
Most Popular Brands of Cleanroom Wipes
PAC (that’s us) is an end-to-end cleanroom supply distributor with locations and warehouses across the Americas. We’re your decontamination specialists. Compare top brands side by side at gotopac.com
Related Posts
-
Class 10,000 Cleanroom & Gowning Area
This cleanroom and adjacent gowning area helped this customer prevent outer contamination from entering classified spaces.
-
Everything You Need to Know About Building A Cleanroom: ISO Class Prefab Hardwall Cleanroom Design
Advantages for Modular Hardwall Design and Construction of ISO 14644-4 Cleanrooms.
-
Cleanroom Cleaning, Gowning, and Maintenance — Procedure, Contamination Control, and ISO Protocol
Proper cleanroom cleaning procedure and maintenance protocol is an immediate, low-cost measure to enhance overall cleanliness, consistency, and contamination control within manufacturing and aseptic cleanrooms. This guide provides a framework for cleanroom management, protocol standards,…
-
New: Texwipe Cleanroom Products
Texwipe offers a variety of dry, pre-saturated, and sterile wipes to meet the strict demands of controlled environments. Texwipe swabs are manufactured to exacting and consistent tolerances with lot coded for traceability and quality control.
-
CleanPro® Softwall Cleanroom Enclosure
This customer needed to enclose a piece of machinery, and CleanPro® was able to provide a solution.
-
CleanPro® Turnkey Cleanroom Install
CleanPro’s turn-key cleanroom solution provided a one-stop, one-contact result for the initial delivery and on-site installation of walls, ceiling grids, electrical systems, flooring, filters, HVAC, and more.