Reprinted with permission by Terra Universal, Inc.
Overview
This document provides a description of the electropolishing (EP) procedures performed by Terra Universal. It also specifies a series of criteria for evaluating the integrity and cleanliness of the electropolished surfaces. Because these electropolishing procedures are followed on all work performed by Terra, the results may be understood as indicative of standard Terra electropolishing, and the specified evaluation criteria may be used as acceptance standards for work performed by Terra.
Introduction
Electropolishing is a reverse plating procedure that entails the electrochemical removal of metal impurities (including carbon, silica, and free iron) from a stainless steel surface. The goal is a smooth surface, devoid of burrs or crevices that attract and trap contaminants.
Prior to electropolishing, parts are mechanically prepared to ensure optimal results. All welds are ground, deburred, and inspected to ensure that all seams are free of pockets or gaps. Finally, selected surfaces are mechanically buffed to a smooth finish.
Next, the part is fitted with electrodes, immersed in an electrolyte solution, and subjected to a direct electrical current. During this electrolytic process, the metallic surface of the anodic part (in this case, stainless steel) is preferentially dissolved ion by ion, yielding a nickel- and chromium-rich surface that protects against metal fatigue or contamination. Optimal results depend on careful control over the current density, the precise chemical composition of the electrolytic solution, the temperature and agitation of the bath, and the duration of current exposure.
Unlike mechanically finished stainless steel, electropolished surfaces feature no fine directional lines and hence offer less friction and surface drag. The chromium-rich surface results in excellent light reflection, yielding a bright, smooth and uniform polish. The images below illustrate the visual difference between EP and non-EP stainless steel surfaces:

EP waste dispenser showing highly reflective, smooth stainless steel.

While shiny, a “grain,” visible on this non-EP stainless steel surface, creates an environment where microparticles are trapped and accumulate.
A Few Words About Stainless Steel
Steel is an iron metal alloy containing other elements that typically include manganese, nickel, chromium, molybdenum, carbon, and silicon. Stainless steel contains a minimum of 50% iron. Some of these other elements influence the hardness and formability of the steel (carbon and silicon, for example), while others have an effect on corrosion resistance (such as chromium).
The amount of chromium present differentiates “regular” steel from “stainless” steel. Chromium that is exposed to oxygen forms a strongly-bonded film of chromium oxide on the stainless steel, blocking the oxygen from damaging the surface, as well as infiltrating the internal structure. Electropolishing increases the surface chromium, thereby promoting formation of this protective film. Non-stainless-steel, with lower levels of chromium, will eventually rust when exposed to moisture and air.
The difference between 304 and 316 stainless steels, the two types that Terra uses, is the ratio of some of the elements present, such as chromium and nickel. 316 stainless steel is a “cleaner” grade of steel, even less susceptible to corrosion than 304.
Lastly, steel comes in mill finishes. Terra uses types 2B or 4, which simply refers to polishing or brushing steps that give the steel a polished aesthetic “finish.” Electropolishing is the last treatment step after the 304 or 316 steel has been manufactured into its final functional form.
Electropolishing Procedures
Terra’s electropolishing is carefully controlled in each of the following areas to ensure the finest results possible:
A. Material Selection
Electropolishing is performed on 304 or 316 stainless steel that has either a 2B or 4 mill finish. This material standard minimizes the presence of sulfide inclusions and other subsurface contaminants and end-grain or large-grain surfaces that can produce a frosty appearance following electropolishing. Incoming material is also inspected for improper annealing (a heat treatment to increase ductility), over-pickling (chemical treatment to remove oxidation), heat scale, and directional roll marks, all of which are accentuated by electropolishing.
B. Precleaning and Post-cleaning
For optimal results, it is important that all surfaces be uniformly exposed to the electrolyte solution, but also that provisions be made to remove all traces of the solution following electropolishing. Failure to remove the solution can result in subsequent outgassing, unacceptable in a clean room environment.
Stainless steel parts intended for electropolishing are designed with these requirements in mind. All welds are carefully inspected to ensure continuous seams, free of pits or gaps where the solution could collect, and all hollow members are drilled to permit effective flushing of the chemical solution after electropolishing.
Because the electropolishing process removes only a very thin surface layer (typically between 0.001″ and 0.0001″), selected surfaces are mechanically buffed, using progressively finer grits to produce the smoothest possible finish.
Following electropolishing, all traces of the electrolyte solution are thoroughly removed from the part, and any hollow cavities are flushed to eliminate the chance of subsequent outgassing. Surfaces are dried and buffed with a soft, non-particulating cloth. Parts are then immediately wrapped in non-shedding material to guard against fingerprints and other surface contaminants while on their journey to the end-user.
C. Process Controls
The more rigorously the electropolishing process variables are controlled, the higher the quality that can be expected in the finished product.
Some of these variables are relatively easy to quantify and monitor, although some variation must be exercised in response to a given sample in order to produce the optimal results.
Electrode Positioning:
Electrode placement is critical to the success of the electropolishing process. Although electrode clamping of objects with a uniform geometrical shape is generally a straightforward task, irregularly shaped objects, which often contain inaccessible cavities or areas exposed to low current densities, present special challenges. Only an experienced technician, equipped with versatile electrode fittings, can ensure optimal results in these situations.
Electrolyte Temperature:
Varying situations may call for varying temperatures, and heating and/or cooling during the electropolishing process may be required.
Electrolyte Agitation:
Only an experienced technician possesses the knowledge of where, when and how to agitate either the electrolyte or the part in order to prevent gassing streaks, flow marks, and similar unacceptable surface anomalies.
Current Duration:
The optimal duration of current exposure depends on the part size and shape. Again, only an experienced electropolisher can control this variable to produce the best results.
Performance Evaluation Parameters
A. Visual Inspection
The first and most obvious test of the effectiveness of the electropolishing is a close visual inspection. In a closely controlled process performed on high-quality material that is adequately prepared for electropolishing, the surface will appear uniformly brilliant, with no detectable pits, streaks, erosion, “frosting,” or other anomalies.
Unlike mechanical buffing, which distorts the surface of the metal and may conceal the material’s true characteristics, electropolishing reveals the imperfections in the structure of the stainless steel. Electropolishing will accentuate any welding flaws, and a non-uniform appearance indicates a high volume of inclusions or a large-grained grade of stainless steel.
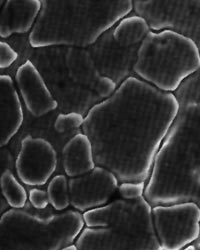
B. Micrographs
A better test of the integrity of the surface is provided by photomicrographs of the surface. Although a highly buffed sample (such as a No. 8 mill mirror finish,) and an electropolished sample may appear equally brilliant to the unaided eye, the differences between the two are apparent when they are viewed under very high magnifications. The sample micrographs below, taken at 1,000X, dramatically illustrate the smooth, featureless surface that results from electropolishing.
This photomicrograph represents the surface of 304 stainless steel with a No. 2B mill finish before Electropolishing. Note that the etched boundaries between the grains are only partially sealed, resulting in a network of sub-surface crevices. Contaminants that lodge in these crevices are protected from contact with cleaning agents leading to subsequent migration of trapped contaminants onto the cleaned surface.
This photomicrograph represents the same No. 4 finish after Electropolishing. The surface may still show some of the abrasive-produced topography to the naked eye, but will now be microscopically featureless with the desired non-contaminating, non-particulating and non-sticking properties.
Related Posts
-
BenchPro™ stainless steel frame workbenches added to PAC product line
Production Automation Corporation is excited to announce that we are now offering BenchPro™ K, D, and A Series workbenches with stainless steel frames. High-quality, durable materials allow you to design and build just the right…
-
FS209E and ISO Cleanroom Standards
Before global cleanroom classifications and standards were adopted by the International Standards Organization (ISO), the U.S. General Service Administration's standards (known as FS209E) were applied virtually worldwide. However, as the need for international standards grew,…
- Eagle Stainless Steel Worktables
Stainless Steel Worktables from Eagle Offers Affordability Combined with Functionality and Durability Eagle HMC offers the industry's widest range of design options for easily configured stainless steel worktables used in industrial applications. These worktables are…
-
Get A Better Stainless Steel Worktable
Stainless Steel Worktables from Eagle Offers Affordability Combined with Functionality and Durability Eagle HMC offers the industry's widest range of design options for easily configured stainless steel worktables used in industrial applications. These worktables are…
-
Choosing a Stainless Steel Table
What is Stainless Steel? Wikipedia describes stainless steel as: inox steel or inox from French “inoxydable“, is defined as a steel alloy with a minimum of 10.5% to 11% chromium content by mass. Stainless steel…
-
Stainless Steel Tables for Sterile Processing
Learn more about the requirements of stainless steel tables during sterile processing.